Differential equation on half-life isotope decay
 Matthew Martinez
Matthew Martinez 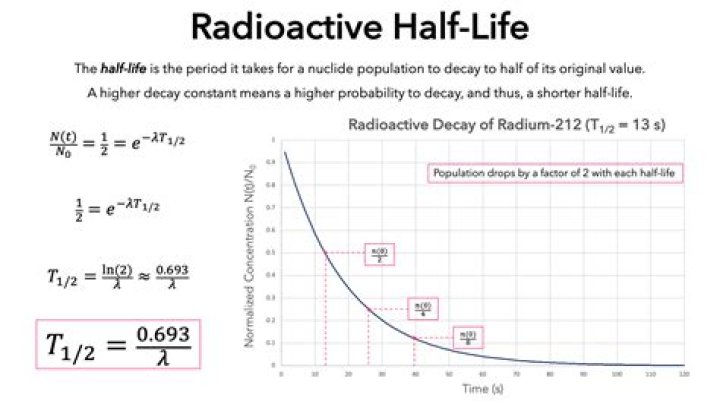
I'm currently working on some Differential Calculus, and I'm having a bit of trouble with the following question.
The half-life of an isotope is 150 years. Use this information to determine the differential equation that describes the mass as a function of time. In other words m' = km where k is a constant and m(t) is the mass after t years.
Use the information given to find k, then solve this equation. Use the solution to determine how long it takes for an initial amount to decay to 15% of the original.
For what I know so far, I know little to nothing on the matter. My professor has failed to elaborate on the subject whatsoever, and I've been googling this to try and answer it.
I think I'm supposed to use this question, but I'm not sure if it's the right one...
(t)/dt%20=%20-k*m(t)Any help?
$\endgroup$1 Answer
$\begingroup$Hint: If you start with 1 unit of the isotope at time $0$, you have $\frac 12$ after $150$ years. Do you know how to solve $m'=km$? That should give you an equation that (with this data) lets you determine $k$. Now put that $k$ into your solution and find the time when the amount is $0.15$.
Added: your equation is the correct differential equation. The solution is $m(t)=m(0)\exp(-kt)$ as the derivative of $\exp(-kt)$ is $-k \exp(-kt)$
$\endgroup$ 4